Kakangari
K-chondrite grouplet, Type 3.6

Fell June 4, 1890
12° 23′ N., 78° 31′ E. After sonic booms were heard, two stones were recovered in Tamilnadu, Salem, Kakangari, India. One stone was broken into pieces while the other, weighing 347 g, was preserved. Most of this meteoriteWork in progress. A solid natural object reaching a planet’s surface from interplanetary space. Solid portion of a meteoroid that survives its fall to Earth, or some other body. Meteorites are classified as stony meteorites, iron meteorites, and stony-iron meteorites. These groups are further divided according to their mineralogy and Click on Term to Read More is accounted for in only four institutions, with the above specimen being obtained through the Natural History Museum in London by an American meteorite dealer.
- an oxidationOxidation and reduction together are called redox (reduction and oxidation) and generally characterized by the transfer of electrons between chemical species, like molecules, atoms or ions, where one species undergoes oxidation, a loss of electrons, while another species undergoes reduction, a gain of electrons. This transfer of electrons between reactants Click on Term to Read More state between H and E chondrites
- a matrix unlike other chondrite groups (enstatite-rich and compositionally similar to the chondrulesRoughly spherical aggregate of coarse crystals formed from the rapid cooling and solidification of a melt at ~1400 ° C. Large numbers of chondrules are found in all chondrites except for the CI group of carbonaceous chondrites. Chondrules are typically 0.5-2 mm in diameter and are usually composed of olivine Click on Term to Read More)
- a high matrix abundance (~70 vol%)
- a high metalElement that readily forms cations and has metallic bonds; sometimes said to be similar to a cation in a cloud of electrons. The metals are one of the three groups of elements as distinguished by their ionization and bonding properties, along with the metalloids and nonmetals. A diagonal line drawn Click on Term to Read More abundance similar to H group ordinary chondrites
- a high troiliteBrass colored non-magnetic mineral of iron sulfide, FeS, found in a variety of meteorites. Click on Term to Read More abundance (10.5 vol%) with depleted chalcophile elementSubstance composed of atoms, each of which has the same atomic number (Z) and chemical properties. The chemical properties of an element are determined by the arrangement of the electrons in the various shells (specified by their quantum number) that surround the nucleus. In a neutral atom, the number of Click on Term to Read More abundances, indicative of large-scale sulfurization
- refractory lithophile and siderophile abundances similar to ordinary chondrites
- chalcophile elements that plot close to R chondrites
- whole-rock O isotopic compositions that plot near the CR chondrites
- chondrule O isotopic compositions that plot with the E chondrites
Kakangari consists of about half chondrules and half chondrule fragments, mostly type-I, FeO-poor just as are EH3 chondrules, and they are FeNi–FeS-rich. In addition, AOAs and refractory material in the form of irregularly shaped CAIs are present. The relative proportion of chondrule textural-types, predominantly porphyritic and non-porphyritic, is unlike that in other chondrite groups; however, the abundance of POP chondrules is most similar to that of ordinary chondrites. Some porphyritic chondrules contain large metal–sulfide intergrowths having ragged outlines, while others contain small metal–sulfide beads within olivines and groundmass. Most chondrules and fragments also have metal–sulfide rims, but some have fine-grained, igneous-textured silicateThe most abundant group of minerals in Earth's crust, the structure of silicates are dominated by the silica tetrahedron, SiO44-, with metal ions occurring between tetrahedra). The mesodesmic bonds of the silicon tetrahedron allow extensive polymerization and silicates are classified according to the amount of linking that occurs between the rims. The abundance of silicate rims in Kakangari is intermediate to that found in the CV and ordinary chondrites. Layered rims are found on some chondrules, implying that they have experienced at least three melting events with two accretionary episodes. Spongy troilite usually forms the final layer on chondrules and fragments.
The matrix mineralogy preserves the thermal history of Kakangari, revealing peak metamorphicRocks that have recrystallized in a solid state due to changes in temperature, pressure, and chemical environment. Click on Term to Read More temperatures of <500°C, with an estimated cooling rate of at least 10 K/m.y. (Nagashima et al., 2015 and references therein). Kakangari matrix is composed of both relatively large clastic grains (enstatiteA mineral that is composed of Mg-rich pyroxene, MgSiO3. It is the magnesium endmember of the pyroxene silicate mineral series - enstatite (MgSiO3) to ferrosilite (FeSiO3). Click on Term to Read More, olivineGroup of silicate minerals, (Mg,Fe)2SiO4, with the compositional endpoints of forsterite (Mg2SiO4) and fayalite (Fe2SiO4). Olivine is commonly found in all chondrites within both the matrix and chondrules, achondrites including most primitive achondrites and some evolved achondrites, in pallasites as large yellow-green crystals (brown when terrestrialized), in the silicate portion Click on Term to Read More, and clinopyroxene) and aggregates of ultrafine-grained, magnesium-rich, crystalline enstatite and olivine, along with lesser amounts of albite, anorthiteRare compositional variety of plagioclase and the calcium end-member of the plagioclase feldspar mineral series with the formula CaAl2Si2O8. Anorthite is found in mafic igneous rocks such as anorthosite. Anorthite is abundant on the Moon and in lunar meteorites. However, anorthite is very rare on Earth since it weathers rapidly Click on Term to Read More, Cr-spinel, troilite, and FeNi-metal (Floss and Stadermann, 2012; Nagashima et al., 2015). The precursor material was likely a mixture of nebular and presolar amorphousMaterial without the regular, ordered structure of crystalline solids. Amorphous substances, like glass, lack a definite repeating pattern in their atomic structures (crystallinity). There may be small regions of order, but, overall there is disorder. Click on Term to Read More or partially crystalline dust. Following thermal metamorphism, which probably caused the destruction of primary presolar grainsMineral grains that formed before our solar system. These tiny crystalline grains are typically found in the fine-grained matrix of chondritic (primitive) meteorites. Most grains probably formed in supernovae or the stellar outflows of red giant (AGB) stars before being incorporated in the molecular cloud from which the solar system Click on Term to Read More, a period of rapid cooling ensued resulting in the production of orthoenstatite and clinoenstatite in the matrix. Chondrules and matrix have similar compositions and both experienced reductionOxidation and reduction together are called redox (reduction and oxidation) and generally characterized by the transfer of electrons between chemical species, like molecules, atoms or ions, where one species undergoes oxidation, a loss of electrons, while another species undergoes reduction, a gain of electrons. This transfer of electrons between reactants Click on Term to Read More processes, likely nebular and parent bodyThe body from which a meteorite or meteoroid was derived prior to its ejection. Some parent bodies were destroyed early in the formation of our Solar System, while others like the asteroid 4-Vesta and Mars are still observable today. Click on Term to Read More, with the chondrules being less reducedOxidation and reduction together are called redox (reduction and oxidation) and generally characterized by the transfer of electrons between chemical species, like molecules, atoms or ions, where one species undergoes oxidation, a loss of electrons, while another species undergoes reduction, a gain of electrons. This transfer of electrons between reactants Click on Term to Read More than matrix silicates. Kakangari chondrules probably formed from material similar to that from which the matrix formed, but reached higher temperatures necessary for complete melting. Solar-wind-implanted He and Ne suggest a residence in a regolithMixture of unconsolidated rocky fragments, soil, dust and other fine granular particles blanketing the surface of a body lacking an atmosphere. Regolith is the product of "gardening" by repeated meteorite impacts, and thermal processes (such as repeated heating and cooling cycles). Click on Term to Read More, with the probability that the meteorite is a brecciaWork in Progress ... A rock that is a mechanical mixture of different minerals and/or rock fragments (clasts). A breccia may also be distinguished by the origin of its clasts: (monomict breccia: monogenetic or monolithologic, and polymict breccia: polygenetic or polylithologic). The proportions of these fragments within the unbrecciated material Click on Term to Read More (Srinivasan and Anders, 1977). Limited aqueous alteration on the parent body produced ferrihydrite and chlorapatite as replacements for kamaciteMore common than taenite, both taenite and kamacite are Ni-Fe alloys found in iron meteorites. Kamacite, α-(Fe,Ni), contains 4-7.5 wt% Ni, and forms large body-centered cubic crystals that appear like broad bands or beam-like structures on the etched surface of a meteorite; its name is derived from the Greek word Click on Term to Read More. Mineralogical anaysis of Kakangari by Nagashima et al. (2015) revealed a majority of FeO-poor type-I chondrules, with only rare FeO-enriched chondrules (not typical type-II). Although silicates in Kakangari show evidence for reduction processes, silicates in E chondrites were reduced to a greater degree than Kakangari as demonstrated by the more Mg-rich chondrules and the higher content of Cr and Ti in the troilite of E chondrites (Berlin et al., 2007). Weisberg et al. (1996) determined that the olivine and pyroxeneA class of silicate (SiO3) minerals that form a solid solution between iron and magnesium and can contain up to 50% calcium. Pyroxenes are important rock forming minerals and critical to understanding igneous processes. For more detailed information, please read the Pyroxene Group article found in the Meteoritics & Classification category. Click on Term to Read More in Kakangari chondrules have a compositional range of Fa0.4-8.0, Fs0.4-10.0, with an average of Fa3.9, Fs5.8. They also found that the composition of LEW 87232 chondrules is Fa1.2, Fs3.3, while data of Zolensky et al. (1989) show the composition of Lea County 002 chondrules to be Fa2.0, Fs4.0. Kakangari and LEW 87232 matrix values for olivine and pyroxene are even more magnesian than those for the chondrules. Values for the K chondrite grouplet as a whole were calculated by Weisberg et al. (1996) using the averages of Kakangari chondrules and matrix in a weighted ratio of 23:77 corresponding to that observed in Kakangari; the resulting values are Fa2.2, Fs4.4. It was suggested that the FeO-rich silicates observed in both the E and K chondrite groups had a common precursor, despite their differences in degree of reduction. In a broader sense, the K chondrites have a combination of properties that do not fit the existing systematics of either the E, O, R, or C chondrites for which isotopic, chemical, and petrologic characteristics vary smoothly relative to their heliocentricCentered around a sun. Our own Solar System is centered around the Sun so that all planets such as Earth orbit around the Sun. Note that 25% of Americans incorrectly believe the Sun revolves around the Earth. Click on Term to Read More distance of formation. For example, the chondrule/matrix ratio is negatively correlated to the oxidation state through the series E>H>L>LL>C3>CM2>CI1, but the K chondrites do not fit into this sequence. RedoxOxidation and reduction together are called redox (reduction and oxidation) and generally characterized by the transfer of electrons between chemical species, like molecules, atoms or ions, where one species undergoes oxidation, a loss of electrons, while another species undergoes reduction, a gain of electrons. This transfer of electrons between reactants Click on Term to Read More analysis suggests that Kakangari contains a unique suite of chondrules (Berlin et al., 2007). It is notable that the models employed by Neumann et al. (2018) indicate a small grain size (0.2 µm) for the precursor material of the acapulcoite–lodraniteRare type of primitive achondrite named after the Lodran meteorite that fell in Pakistan in 1868. Initially, lodranites were grouped with the stony-iron meteorites because they contain silicates (olivine, orthopyroxene, and minor plagioclase) and Fe-Ni metal in nearly equal proportions. However, since discovery of the closely related acapulcoite group, lodranites Click on Term to Read More (AL) parent body, consistent with a large matrix component. Considering the high matrix volume present in K chondrites, along with the mineralogical and geochemical data for this rare group, they speculated that the K chondrites might represent the thin chondritic outer layer of the AL parent body. Generally, the O-isotope composition of the bulk chondrules in Kakangari are 16O-poor and plot along the terrestrial fractionationConcentration or separation of one mineral, element, or isotope from an initially homogeneous system. Fractionation can occur as a mass-dependent or mass-independent process. Click on Term to Read More line on an oxygenElement that makes up 20.95 vol. % of the Earth's atmosphere at ground level, 89 wt. % of seawater and 46.6 wt. % (94 vol. %) of Earth's crust. It appears to be the third most abundant element in the universe (after H and He), but has an abundance only Click on Term to Read More three-isotope diagram (ave. olivine: Δ17O +0.0 [±0.8] ‰; ave. low-Ca pyroxene: +0.2 [±0.9] ‰), while that of the matrix olivine and enstatite grains are slightly 16O-enriched by 2.6‰ (Nagashima et al., 2011, 2015). Analyses show that the matrix grains actually exhibit a bimodal distribution with a majority that are 16O-rich (Δ17O ~ –23.5 [±2.9] ‰) and others that are 16O-poor (Δ17O –0.1 [±1.7] ‰). In accordance with these values, the O-isotope composition of olivines composing a coarse-grained igneous rim on a porphyritic chondrule is 16O-rich (Δ17O ~ –24‰). As indicated by the O-isotopic data, Nagashima et al. (2015) deduced that Kakangari chondrule and matrix components likely formed from the same nebular reservoir, and that the observed 16O-enrichment in the bulk matrix compared to the 16O-poor bulk chondrules is due to the presence of 16O-rich grains in the matrix.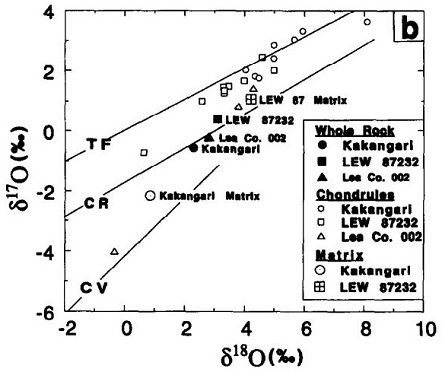
Diagram credit: Weisberg et al., GCA, vol. 60, p. 9 (1996)
‘The K (Kakangari) chondrite grouplet’
(https://doi.org/10.1016/S0016-7037(96)00233-5) A NanoSIMS study of Kakangari conducted by Floss and Stadermann (2009, 2012) revealed a single C-anomalous grain (13C-rich), probably SiC from AGB starsStars on the Asymptotic Giant Branch, which represents a late stage of stellar evolution that all stars with initial masses < 8 Msun go through. At this late stage of stellar evolution, gas and dust are lifted off the stellar surface by massive winds that transfer material to the interstellar Click on Term to Read More, corresponding to an abundance of ~4 ppmParts per million (106). Click on Term to Read More. Kakangari also contains relatively low abundances of presolar silicate/oxide grains (< ~5 ppm). Furthermore, there is a complete lack of both O-anomalous presolar grains and amorphous silicates in the matrix. These findings attest to the fact that Kakangari experienced secondary processing, probably on the parent body, and thus cannot be considered a primitive meteorite. The fusion crustMelted exterior of a meteorite that forms when it passes through Earth’s atmosphere. Friction with the air will raise a meteorite’s surface temperature upwards of 4800 K (8180 °F) and will melt (ablate) the surface minerals and flow backwards over the surface as shown in the Lafayette meteorite photograph below. Click on Term to Read More of Kakangari is described as dull and dark brown, exhibiting a close textured, locally ribbed and netted texture (S. Ghosh and A. Dube, 1999). The photos above show a 1.15 g surface fragment of Kakangari dispalying both the fusion-crusted side and the interior.

Photo courtesy of Geological Survey of India, Catalogue Series No. 3, S. Ghosh and A. Dube (© 1999)







