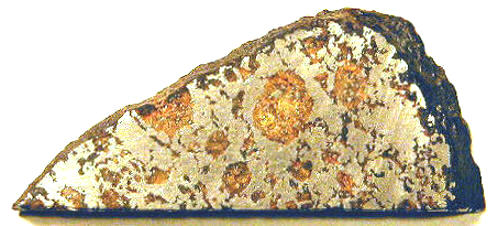
NWA 176
Iron with silicateThe most abundant group of minerals in Earth's crust, the structure of silicates are dominated by the silica tetrahedron, SiO44-, with metal ions occurring between tetrahedra). The mesodesmic bonds of the silicon tetrahedron allow extensive polymerization and silicates are classified according to the amount of linking that occurs between the inclusions, ungroupedModifying term used to describe meteorites that are mineralogically and/or chemically unique and defy classification into the group or sub-group they most closely resemble. Some examples include Ungrouped Achondrite (achondrite-ung), Ungrouped Chondrite (chondrite-ung), Ungrouped Iron (iron-ung), and Ungrouped Carbonaceous (C-ung). Click on Term to Read More
(CO–CV clan related)
Found 1999
no coordinates recorded A rare Saharan iron meteoriteIron meteorites consist mostly of metallic iron alloyed with typically between ~5 to ~30 wt% nickel. The main metal phases are kamacite α-(Fe, Ni) and taenite y-(Fe, Ni). Based on their group classification, they may also contain a small weight percentage of one or more of the following minerals: • Click on Term to Read More weighing 2 kg was found near the border of Morocco and Algeria. The mass was obtained by G. Cintron of Island Meteorites, and a portion was sent for study to several institutions, including the Hawaii Institute of Geophysics & Planetology, the Institute of Geophysics and Planetary Physics in Los Angeles, the Enrico Fermi Institute in Chicago, the Universitat Bern in Switzerland, and Washington University in St. Louis. This meteoriteWork in progress. A solid natural object reaching a planet’s surface from interplanetary space. Solid portion of a meteoroid that survives its fall to Earth, or some other body. Meteorites are classified as stony meteorites, iron meteorites, and stony-iron meteorites. These groups are further divided according to their mineralogy and Click on Term to Read More contains sub-mm- to cm-sized, rounded, greenish-yellow, silicate inclusions dispersed throughout the metalElement that readily forms cations and has metallic bonds; sometimes said to be similar to a cation in a cloud of electrons. The metals are one of the three groups of elements as distinguished by their ionization and bonding properties, along with the metalloids and nonmetals. A diagonal line drawn Click on Term to Read More host constituting ~44 vol%. Some of the silicates are aligned along shear planes that were probably created during an impact event.
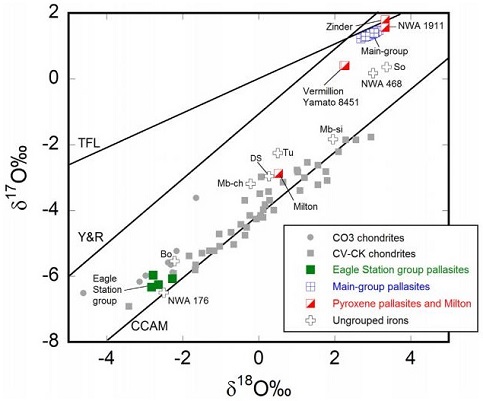
Diagram credit: Greenwood et al., Chemie der Erde, vol. 77, p. 21, (2017)
‘Melting and differentiationA process by which a generally homogeneous chondritic body containing mostly metal, silicates and sulfides will melt and form distinct (differentiated) layers of different densities. When the melting process continues for a long enough period of time, the once chondritic body will re-partition into layers of different composition including Click on Term to Read More of early-formed asteroids: The perspective from high precision oxygen isotope studies’
(open access: http://dx.doi.org/10.1016/j.chemer.2016.09.005) Chromium vs. Oxygen Isotope Plot
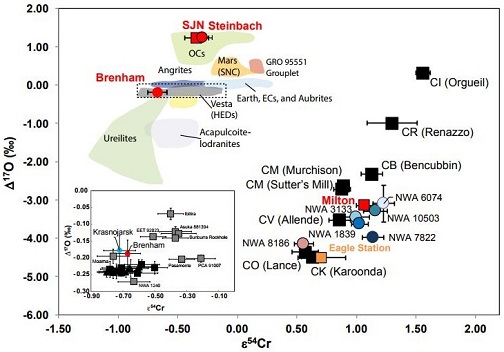
click on diagram for a magnified view Diagram credit: Sanborn et al., 49th LPSC, #1780 (2018) Northwest Africa 176 and Bocaiuva, along with the Eagle Station group pallasites, are resolved from the main-group pallasites and the major iron groups by their higher Ge/Ga ratios, higher Cu and Ir contents, and lower Au, As, and Sb contents. For those few irons that do have Ge/Ga ratios similar to NWA 176, in particular, the irons of group IIF and certain ungrouped irons such as the silicated iron Mbosi, their elemental abundance ratios rule against a genetic relationship with NWA 176 and its relatives. Nevertheless, it is likely they all originated from similar chondritic precursor material in the carbonaceous reservoir beyond Jupiter. A multi-stage formation history is proposed for NWA 176 in which an initial impact generated enough heat (~1100°C) to form a melt pool. This was followed by gravitational differentiation that was sustained above ~500°C for a significant time. Differentiation resulted in a lower metallic layer with a cumulateIgneous rock composed of crystals that have grown and accumulated (often by gravitational settling) in a cooling magma chamber. Click on Term to Read More silicate layer above. A subsequent impact event shattered the silicate layer and mobilized metal forcing it into existing cracks in the silicate layer, while initiating a rapid cooling phase at a rate of 1000°C/m.y. This cooling data would be consistent with a small body of only a few km in radius, or possibly reflects the breakup phase of a much larger object. Extended annealing at depth led to the rounding of the corners on silicate grains, a thermodynamic process acting to minimize the surface energy. Several factors support this origin rather than the core–mantleMain silicate-rich zone within a planet between the crust and metallic core. The mantle accounts for 82% of Earth's volume and is composed of silicate minerals rich in Mg. The temperature of the mantle can be as high as 3,700 °C. Heat generated in the core causes convection currents in Click on Term to Read More interface origin commonly envisioned for the main-group pallasites. The nearly chondritic silicate composition with its relatively low proportion of olivineGroup of silicate minerals, (Mg,Fe)2SiO4, with the compositional endpoints of forsterite (Mg2SiO4) and fayalite (Fe2SiO4). Olivine is commonly found in all chondrites within both the matrix and chondrules, achondrites including most primitive achondrites and some evolved achondrites, in pallasites as large yellow-green crystals (brown when terrestrialized), in the silicate portion Click on Term to Read More is more consistent with an impact-melt model than for a core–mantle interface origin. Moreover, the heat-generating radiogenic isotopes present in the later-forming carbonaceous chondriteCarbonaceous chondrites represent the most primitive rock samples of our solar system. This rare (less than 5% of all meteorite falls) class of meteorites are a time capsule from the earliest days in the formation of our solar system. They are divided into the following compositional groups that, other than Click on Term to Read More parent bodies would be insufficient to produce melts that could form pallasitic compositions. Furthermore, the shear forces that produced the deformation features, as well as the loss of alkali volatiles, can be reconciled through impact mixing processes. Finally, the low Ir content of NWA 176 and its relatives is not consistent with fractional crystallizationA crystallization process in which minerals crystallizing from a magma are isolated from contact with the liquid. It is a key process in the formation of igneous rocks during the process of magmatic differentiation. Also known as crystal fractionation. Click on Term to Read More processes necessary for a core–mantle origin. One possible alternative origin proposed for this meteorite is that the precursor was a metal-rich, highly metamorphosed, chondritic asteroid. Further details about possible origins of NWA 176 can be found on the Allende page. Northwest Africa 176 has an absolute I–Xe age, calculated relative to Shallowater (4.5623 [±0.0004] b.y.), of ~4.544 (±0.007) b.y., indicating a relatively late formation, probably through impact processes (Bogard and Garrison, 2009). The meteorite has a remarkably high Ar–Ar age of 4.524 (±0.013) b.y. The CRE age of 41 (±12) m.y. calculated for NWA 176 is low compared to other iron meteorites. A portion of the information above was gleaned from the paper ‘Bocaiuva—A Silicate-Inclusion Bearing Iron Meteorite Related To The Eagle-Station Pallasites’ (Malvin, Wasson, Clayton, Mayeda, & Curvello), MeteoriticsScience involved in the study of meteorites and related materials. Meteoritics are closely connected to cosmochemistry, mineralogy and geochemistry. A scientist that specializes in meteoritics is called a meteoriticist. Click on Term to Read More, vol. 20, #2, Part 1 (1985). The specimen of NWA 176 pictured above is an 8.07 g partial slice displaying rounded silicate inclusions. It’s fresh dark fusion crustMelted exterior of a meteorite that forms when it passes through Earth’s atmosphere. Friction with the air will raise a meteorite’s surface temperature upwards of 4800 K (8180 °F) and will melt (ablate) the surface minerals and flow backwards over the surface as shown in the Lafayette meteorite photograph below. Click on Term to Read More attests to a recent arrival.