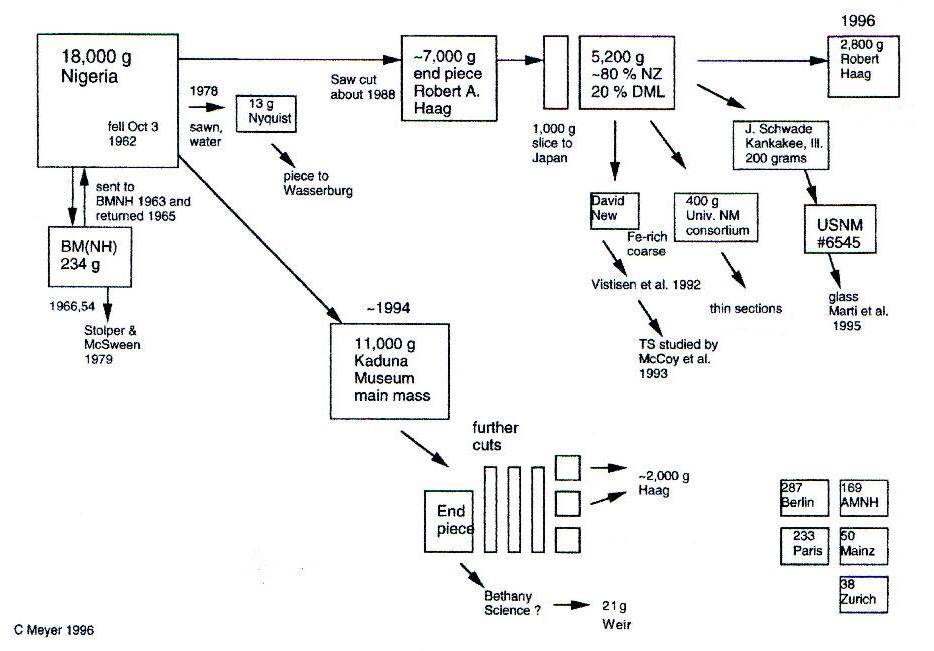
Zagami
Martian ShergottiteIgneous stony meteorite with a Martian origin consisting mainly of plagioclase (or a shocked glass of plagioclase composition) and pyroxene. They are the most abundant type of SNC meteorites and the type member is the Shergotty meteorite, which fell in India in 1865. Shergottites are igneous rocks of volcanic or Click on Term to Read More
low-Al basaltBasalt is the most common extrusive igneous rock on the terrestrial planets. For example, more than 90% of all volcanic rock on Earth is basalt. The term basalt is applied to most low viscosity dark silicate lavas, regardless of composition. Basalt is a mafic, extrusive and fine grained igneous rock Click on Term to Read More ∗
(enriched, maficOne of the two broad categories of silicate minerals, the other being felsic, based on its magnesium (Mg) and/or iron (Fe) content. Mafic indicates silicate minerals that are predominantly comprised of Mg and/or Fe.The term is derived from those major constituents: Magnesium + Ferrum (Latin for iron) + ic (having Click on Term to Read More)
click on photo for a magnified view Fell October 3, 1962
11° 44′ N., 7° 5′ E. An eyewitness account of this meteoriteWork in progress. A solid natural object reaching a planet’s surface from interplanetary space. Solid portion of a meteoroid that survives its fall to Earth, or some other body. Meteorites are classified as stony meteorites, iron meteorites, and stony-iron meteorites. These groups are further divided according to their mineralogy and Click on Term to Read More fallMeteorite seen to fall. Such meteorites are usually collected soon after falling and are not affected by terrestrial weathering (Weathering = 0). Beginning in 2014 (date needs confirmation), the NomComm adopted the use of the terms "probable fall" and "confirmed fall" to provide better insight into the meteorite's history. If Click on Term to Read More was provided to Robert ‘Meteoriteman’ Haag at the actual site. Haag related in his Meteorite Field Guide (1991) that a man who was chasing cows out of his cornfield near the village of Zagami Rock, Nigeria, suddenly heard a loud explosion and was buffetted by a pressure wavePropagating disturbance which transmits energy from one point to another without physically transporting the oscillating quantity. A wave is characterized by wavelength and frequency.. Seconds later there was a puff of smoke and a thud about ten feet away. Fearing that an artillery shell had landed, the man waited for a few minutes before approaching the two-foot hole. The black rock was recovered and placed in a museum in Kaduna.
A calcium-rich, basaltic rock, having a pre-atmospheric size of ≤0.5 m and a weight of at least 150 kg (Eugster et al., 2002), had just completed a 3-million-year journey from Mars. Following an atmospheric ablationGradual removal of the successive surface layers of a material through various processes. • The gradual removal and loss of meteoritic material by heating and vaporization as the meteoroid experiences frictional melting during its passage through the atmosphere. The resulting plasma ablates the meteor and, in cases where a meteor Click on Term to Read More of more than 90%, this rare martian rock was reducedOxidation and reduction together are called redox (reduction and oxidation) and generally characterized by the transfer of electrons between chemical species, like molecules, atoms or ions, where one species undergoes oxidation, a loss of electrons, while another species undergoes reduction, a gain of electrons. This transfer of electrons between reactants Click on Term to Read More to a single 18 kg mass. Evidence supporting a martian origin includes the following:
- a young crystallizationPhysical or chemical process or action that results in the formation of regularly-shaped, -sized, and -patterned solid forms known as crystals. Click on Term to Read More age of only 180 m.y. (however, this age might instead represent a shock resetting event accompanied by maskelyniteNatural glass composed of isotropic plagioclase produced during shock metamorphism (not melting) at pressures of ~30 GPa. Maskelynite is commonly found in shergottites though also found in some ordinary chondrites, HED and lunar meteorites. It is also found in association with meteorite impact craters and crater ejecta. Named after British Click on Term to Read More production [Jagoutz and Dreibus, 2002; Eugster et al., 1997], or perhaps metasomatic homogenation affecting the radiometric chonometers [Albarède et al., 2008], and the actual crystallization age might be 4.1 b.y.)
- a mineralInorganic substance that is (1) naturally occurring (but does not have a biologic or man-made origin) and formed by physical (not biological) forces with a (2) defined chemical composition of limited variation, has a (3) distinctive set of of physical properties including being a solid, and has a (4) homogeneous Click on Term to Read More composition in which maskelynites contain ~90–3600 ppmParts per million (106). Click on Term to Read More water, for which the relatively high D/H ratios are consistent with maskelynite interacting with groundwater that had exchanged with a D-enriched martian atmosphere (Tucker et al., 2014).
- the presence of trapped martian atmospheric gases, with isotopic percentages matching those measured by the Viking and Pathfinder missions.
- features of a weak gravityAttractive force between all matter - one of the four fundamental forces. Click on Term to Read More field acting on the crystallizing minerals.
- residual magnetic properties.
- unique Mn/Fe ratios in pyroxeneA class of silicate (SiO3) minerals that form a solid solution between iron and magnesium and can contain up to 50% calcium. Pyroxenes are important rock forming minerals and critical to understanding igneous processes. For more detailed information, please read the Pyroxene Group article found in the Meteoritics & Classification category. Click on Term to Read More.
- a unique O-isotopic signature common to all SNC meteorites, but distinct from other meteorite classes.
- a positive match between basaltic shergottitesIgneous stony meteorite with a Martian origin consisting mainly of plagioclase (or a shocked glass of plagioclase composition) and pyroxene. They are the most abundant type of SNC meteorites and the type member is the Shergotty meteorite, which fell in India in 1865. Shergottites are igneous rocks of volcanic or Click on Term to Read More (EETA79001B and QUE 94201) and a rock from the martian plains at Meridiani Planum, named ‘Bounce Rock’, from analyses of a suite of sophisticated instruments employed by the Mars Exploration Rover.
The martian shergottite group was previously divided into two distinct subgroups, the basaltic and ‘lherzolitic’ subgroups. In actuality, the ‘lherzolitic’ shergottites do not contain the minimum abundances of olivineGroup of silicate minerals, (Mg,Fe)2SiO4, with the compositional endpoints of forsterite (Mg2SiO4) and fayalite (Fe2SiO4). Olivine is commonly found in all chondrites within both the matrix and chondrules, achondrites including most primitive achondrites and some evolved achondrites, in pallasites as large yellow-green crystals (brown when terrestrialized), in the silicate portion Click on Term to Read More or orthopyroxeneOrthorhombic, low-Ca pyroxene common in chondrites. Its compositional range runs from all Mg-rich enstatite, MgSiO3 to Fe-rich ferrosilite, FeSiO3. These end-members form an almost complete solid solution where Mg2+ substitutes for Fe2+ up to about 90 mol. % and Ca substitutes no more than ~5 mol. % (higher Ca2+ contents occur Click on Term to Read More as those established for terrestrial lherzolites. Since there was no known petrologic relationship existing between the basaltic and ‘lherzolitic’ shergottite subgroups, and these groups are resolved from each other on an O-isotope plot, the use of the term lherzolites was proposed by Eugster and Polnau (1997) to represent this unique group of martian meteorites. Thereafter, in an effort to resolve the discrepencies that exist between the official IUGS definition of lherzolites and the application of that term to the varied group of ‘lherzolitic’ shergottites, Mikouchi (2009) addressed the need for changing the name of the ‘lherzolitic’ shergottites to one that is more consistent and more broadly applicable. Since a texturally-based nomenclature is already employed for some shergottite subgroups, e.g., olivine-phyric, it was suggested that the term ‘pyroxene-oikocrystic’ shergottites would be an appropriate name with which to encompass all of the various martian ‘lherzolitic’ shergottites that exist in the worldwide collections. This would include intermediate, enriched, and postulated depleted ‘lherzolitic’ shergottites, as reflected by a geochemical classification scheme. More recently, in an effort to rectify the discrepencies that exist in martian meteoriteOver 30 of the meteorites found on Earth almost certainly came from Mars (see http://www.imca.cc/mars/martian-meteorites.htm and http://www2.jpl.nasa.gov/snc/). All but one belongs to the group known as SNC meteorites, which includes the shergottites, nakhlites, and chassignites. SNC meteorites contain minerals that crystallized within the past 1.35 to 0.15 Ga, making them Click on Term to Read More nomenclature, the textural term ‘poikilitic’ was proposed by Walton et al. (2012) to apply to those meteorites previously referred to as ‘lherzolitic’ shergottites, which is to be used along with additional descriptive terms for the bulk major elementSubstance composed of atoms, each of which has the same atomic number (Z) and chemical properties. The chemical properties of an element are determined by the arrangement of the electrons in the various shells (specified by their quantum number) that surround the nucleus. In a neutral atom, the number of Click on Term to Read More composition (based on Mg/(Mg + Fe); mafic, permafic, or ultramaficTerm used for silicate minerals with cations predominantly Mg and/or Fe. Mafic minerals are dominated by plagioclase and pyroxene, and also contain smaller amounts of olivine. Click on Term to Read More) and the trace element content (depleted, intermediate, or enriched) of a meteorite.
The division of the remaining shergottites have also undergone a revision:
- An olivine-poor basalt subgroup comprising those meteorites with a volcanicIgneous rock that forms from cooling magma on the surface of a planet or asteroid. origin derived from a fractionated magmaMolten silicate (rock) beneath the surface of a planetary body or moon. When it reaches the surface, magma is called lava. Click on Term to Read More and consisting primarily of the clinopyroxenes pigeoniteLow-Ca clinopyroxene, (Ca,Mg,Fe)SiO3, found as a major mineral in eucrites and shergottites. In order to be considered pigeonite, the clinopyroxene must contain 5 to 20 mol % of calcium (Wo5 - 20). Chondrites of petrologic types 4 and below contain significant low-Ca clinopyroxene. During metamorphism to higher temperatures, all existing and augiteHigh-Ca clinopyroxene, (Ca,Mg,Fe)SiO3, that occurs in many igneous rocks, particularly those of basaltic composition. In order to be considered augite, the clinopyroxene must contain 20 to 45 mol % of calcium (Wo20 - 45). An important and unique Martian meteorite is NWA 8159, that has been classified as an augite basalt. Click on Term to Read More, in addition to having a high abundance of feldspathic glass (actually, most basaltic shergottites are more accurately termed komatiites based on their low plagioclaseAlso referred to as the plagioclase feldspar series. Plagioclase is a common rock-forming series of feldspar minerals containing a continuous solid solution of calcium and sodium: (Na1-x,Cax)(Alx+1,Si1-x)Si2O8 where x = 0 to 1. The Ca-rich end-member is called anorthite (pure anorthite has formula: CaAl2Si2O8) and the Na-rich end-member is albite Click on Term to Read More content and depleted trace element composition).
- A subgroup with olivine-porphyritic textures. The name picritic shergottite was suggested for this new subgroup by Barrat et al. (2002), while the name olivine-phyric shergottite was suggested by Goodrich (2002). Goodrich suggests that the term picritic shergottite implies certain petrogenetic characteristics, such as mixing of two compositionally distinct magma sources, which is not necessarily the case for all members of this new subgroup; therefore the purely descriptive term olivine-phyric is favored.
- Recently, several shergottites with both olivine and orthopyroxene megacrysts have been identified, which has led to the proposal of a new shergottite subgroup—the olivine–orthopyroxene-phyric shergottites.
Although ALH 84001 is an orthopyroxeniteA rock composed primarily of orthopyroxene. Non-terrestrial orthopyoxenites include diogenites and a single martian meteorite, ALH 84001, that was found in the Allan Hills region of Antarctica in 1984. ALH 84001 is a cumulate rock consisting of 97% coarse-grained, Mg-rich orthopyroxene, with small amounts of plagioclase, chromite, and carbonate. It Click on Term to Read More, and as such was characterized by the Planetary Chemistry Laboratory at Washington University as a subgroup of the nakhlites, its parental source magma has a composition that is consistent with the same mixtures of depleted and enriched REEOften abbreviated as “REE”, these 16 elements include (preceded by their atomic numbers): 21 scandium (Sc), 39 Yttrium (Y) and the 14 elements that comprise the lanthanides excluding 61 Promethium, an extremely rare and radioactive element. These elements show closely related geochemical behaviors associated with their filled 4f atomic orbital. Click on Term to Read More end-member components that are used in a geochemical classification of the shergottites (Lapen et al., 2012). It was determined that the source magma of ALH 84001 contained a higher proportion of the enriched REE component than all other shergottites studied thus far. Therefore, ALH 84001 may be most appropriately classified as a subgroup of the shergottites.
The shergottite group as a whole contains a large proportion of plagioclase feldsparAn alumino-silicate mineral containing a solid solution of calcium, sodium and potassium. Over half the Earth’s crust is composed of feldspars and due to their abundance, feldspars are used in the classification of igneous rocks. A more complete explanation can be found on the feldspar group page. Click on Term to Read More, which was shocked to pressures of ~30 GPa creating maskelynite intergrowths. The presence of other high-pressure silicaSilicon dioxide, SiO2. polymorphs, including cristobaliteHigh temperature polymorph of silicon dioxide (SiO2). Has the same chemical composition as coesite, stishovite, seifertite and tridymite but possesses a different crystal structure. This silica group mineral occurs in terrestrial volcanic rocks, martian and lunar meteorites, chondrites and impact glasses like Libyan Desert Glass. Cristobalite has a very open Click on Term to Read More, high-pressure glass, stishoviteDense, high-pressure phase of quartz; so far identified only in shock-metamorphosed, quartz-bearing rocks from meteorite impact craters. Stishovite was synthesized in 1961 before it was discovered at Meteor Crater, Arizona. Its structure consists of parallel chains of single SiO6 octahedra. The octahedra are on their sides, sharing opposing edges. Image Click on Term to Read More, post-stishovite (only identified by cathodoluminescenceEmission of visible light in response to electron bombardment. Click on Term to Read More techniques), and the newly discovered mineral liebermannite (Ma et al., 2015), suggests that localized regions experienced higher pressures of >40 GPa. The tiny, darkened melt veins in Zagami were formed as a result of shock-induced shear deformation, perhaps at low shock pressures (Bogert et al., 2003).
The Zagami meteorite includes a range of lithologies formed through progressive fractional crystallizationA crystallization process in which minerals crystallizing from a magma are isolated from contact with the liquid. It is a key process in the formation of igneous rocks during the process of magmatic differentiation. Also known as crystal fractionation. Click on Term to Read More of the magma, which reflects increasing enrichment of FeO and incompatible elements (McCoy et al., 1999). Most prevalent (~80%) is the early-crystallizing normal lithology (NZ), composed of both fine-grained (FG) and coarse-grained (CG) components, which are crossed by shock-melt veins. The parent melt of the fine-grained lithology may have inherited a larger abundance of pre-existing pyroxene nuclei than did the coarse-grained lithology parent melt (Nyquist et al, 2006). Next in order to crystallize, constituting most of the remaining evolved, incompatible element-enriched rock, was an FeO-enriched component, the dark-mottled lithology (DML). The DML has been found to contain a small component (~10%) of late-stage melt pockets that are enriched in fayalitePure* iron end-member (Fe2SiO4) of the olivine solid solution series and an important mineral in meteorites. When iron (Fe) is completely substituted by magnesium, it yields the the pure Mg-olivine end-member, forsterite (Mg2SiO4). The various Fe and Mg substitutions between these two end-members are described based on their forsteritic (Fo) Click on Term to Read More, phosphates, sulfides, oxides (e.g., ZrO2), and mesostases, and which contain a martian atmosphere component. However, these olivine-rich melt pockets contain an anomalous Sr-isotopic signature inconsistent with very late-stage fractional crystallization, and may instead reflect the infiltration of a highly evolved magma into a nearly solidified body (Misawa et al., 2012). The phosphates in these melt pockets consist primarily of whitlockite and contain ~5000 ppm martian water, reflecting either a very dry magma source or efficient outgassing. One very large, highly-evolved, olivine-rich melt pocket located in the DML component was named after its finder, David New (DN).
A large apatite grain in the DN lithology was found to contain 3000–4000 ppm water with a high D/H ratio (Watson et al., 1994). Maltsev et al. (2015) extracted water from Zagami during stepwise heating, and the highest Δ17O value (between 200–400°C) was found to be 0.1‰. This value is lower than that calculated for Zagami bulk rock (0.33‰), but higher than the terrestrial value; such variability is consistent with multiple isotopically distinct water reservoirs on Mars. Boctor et al. (2014) measured the water content and the H-isotopic composition of maskelynites from Zagami. They found that a wide range exists, ~12–99 ppm, having a δD of ~579–2532‰. In subsequent studies, Tucker et al. (2014) found water contents of ~90–3600 ppm having a δD range of ~0–+1100‰. Further maskelynite analyses by Tucker et al. (2015) found water contents of 93–223 ppm with a δD range of –93–+849‰.
Similar to other highly shocked martian meteorites, Zagami contains a significant concentration of martian atmospheric Ar within melt pockets (ave. 19.7 ppbParts per billion (109). Click on Term to Read More), with a minor component present within shock veins (ave. 1.2 ppb). The favored scenario explaining the existence of this trapped gas component within the late-stage melt pockets argues for the initial introduction of martian atmospheric gas into pre-existing cracks and pores. Following the passage of a shock waveAbrupt perturbation in the temperature, pressure and density of a solid, liquid or gas, that propagates faster than the speed of sound., sudden decompression and pressure release occurred creating bubbles within sub-mm-sized, localized melt pockets. Thereafter, as pressures became equilibrated, the trapped atmospheric gases migrated into the vesicles of the melt phase from the surrounding cracks and pores (Walton et al., 2007). Zagami is very similar to terrestrial basalts in even minor and trace element contents. Through studies involving O, N, and Cr isotopic compositions of martian meteorites, along with bulk planetary Fe/Si ratios, it is proposed that Mars accreted from early Solar SystemThe Sun and set of objects orbiting around it including planets and their moons and rings, asteroids, comets, and meteoroids. material comprising enstatiteA mineral that is composed of Mg-rich pyroxene, MgSiO3. It is the magnesium endmember of the pyroxene silicate mineral series - enstatite (MgSiO3) to ferrosilite (FeSiO3). Click on Term to Read More and ordinary chondriteWork in Progress Ordinary chondrites (OCs) are the largest meteorite clan, comprising approximately 87% of the global collection and 78% of all falls (Meteoritical Society database 2018)1. Meteorites & the Early Solar System: page 581 section 6.1 OC of type 5 or 6 with an apparent shock stage of S1, Click on Term to Read More material in a ratio of 74:26, in accord with a coreIn the context of planetary formation, the core is the central region of a large differentiated asteroid, planet or moon and made up of denser materials than the surrounding mantle and crust. For example, the cores of the Earth, the terrestrial planets and differentiated asteroids are rich in metallic iron-nickel. Click on Term to Read More that contains 6.7 wt% Si (Mohapatra and Murty, 2003).
The clinopyroxene crystals in Zagami are composed of pigeonite and augite with Mg-rich cores, which suggests that initial crystallization began inside a slowly cooling, fractionating magma chamber (0.1–0.5°C/hr) at a depth of ~7–15 km. This was followed by a nucleation hiatus in which pyroxene grain-size increased at the expense of the smallest grains. The resumption of crystal nucleation and growth occurred during magma ascent and subsequent eruption onto the surface. Preliminary findings indicate that both olivine and pyroxene grains have a preferred orientation (Stephen et al., 2010), It was shown by Becker et al. (2011) that the weaker foliation associated with the coarse-grained pyroxene of the NZ lithology, in contrast to the fine-grained lithology, was not formed by a strong directional flow; perhaps more consistent with a shallow intrusiveRefers to igneous rocks that crystallized underground. Click on Term to Read More.
In a study of the light lithophile elementElement that tends to be concentrated in the silicate phase, e.g., B, O, halogens, alkali earths, alkali metals, Al, Si, Sc, Ti, V, Cr, Mn, Y, Zr, Nb, REE, Hf, Ta, W, Th, and U. concentrations in Zagami pyroxenes (Herd et al., 2005), particularly that of the incompatible element Li, it was found that Li exhibited significant zoning, decreasing from core to rim by 51% (and by 75% in Shergotty). This zoning has been attributed to possible postcrystallization partitioningThe tendency of elements to prefer one mineral phase relative to another or to preferentially enter the solid or remain in the liquid during crystallization. Click on Term to Read More of Li into magmatic water, which was subsequently degassed; however, shock-induced diffusionMovement of particles from higher chemical potential to lower chemical potential (chemical potential can in most cases of diffusion be represented by a change in concentration). Diffusion, the spontaneous spreading of matter (particles), heat, or momentum, is one type of transport phenomena. Because diffusion is thermally activated, coefficients for diffusion Click on Term to Read More from the rims remains another possible alternative. In a related study of the Li systematics in NWA 480, Beck et al. (2004) found that Li in the pyroxenes reveals a large isotopic variation from core to rim, but that it also maintains a constant concentration. They suggest that these compositional trends for Li reflect the loss of Li (i.e., mass-fractionation) from the crystallizing melt through the degassing of water-rich fluids, a process similar to that proposed to have occurred in Zagami and Shergotty.
The Os–Nd isotopic variations in Zagami and other shergottites suggest that a single parent magma assimilated an LREE-enriched crustal component before cooling (Brandon et al., 2011). On the other hand, the failure of crustal assimilation models to realistically reproduce the isotopic variation observed among shergottites has led some investigators to consider alternative models involving source mantleMain silicate-rich zone within a planet between the crust and metallic core. The mantle accounts for 82% of Earth's volume and is composed of silicate minerals rich in Mg. The temperature of the mantle can be as high as 3,700 °C. Heat generated in the core causes convection currents in Click on Term to Read More heterogeneity. The enriched (upper mantle, 200–450 km deep) and depleted (either upper or deeper mantle) shergottite end-members are thought to reflect crystallization and mixing of cumulates with trapped residual melt at various depths within a 2,000-km-deep magma oceanCompletely molten surfaces of terrestrial planets or moons that formed soon after accretion. Samples returned by the Apollo missions provide evidence of a lunar magma ocean, crystallization of which produced a stratified Moon with a low-density crust formed by accumulation of the mineral plagioclase overlying a higher density mantle of Click on Term to Read More, following 89–93% crystallization. These conditions are consistent with an origin in a typical Tharsis-type, volcanic magma chamber, such as Olympus MonsLarge area of high relief; mountain (pl. montes). Click on Term to Read More (see photos below). Some studies suggest that a later period of rapid cooling in a lavaHot molten or semifluid rock derived from a volcano or surface fissure from a differentiated and magmatically active parent body. Click on Term to Read More flow 10 m thick produced the Fe-rich rims on the pyroxene cores and created the partial flow alignment of the crystals (exhibited in the NZ lithology only).
OLD VS. YOUNG CRYSTALLIZATION AGE
Using U–Pb, Rb–Sr, Sm–Nd, and Lu–Hf isotopic systematics, it was determined that Zagami formed from one of the most highly fractionated and evolved magma sources on Mars very early in Solar SystemDefinable part of the universe that can be open, closed, or isolated. An open system exchanges both matter and energy with its surroundings. A closed system can only exchange energy with its surroundings; it has walls through which heat can pass. An isolated system cannot exchange energy or matter with history that existed ~4.558 b.y. ago. It was further ascertained through concordant isotopic chronometers that crystallization of Zagami from the molten state took place as recently as ~166 m.y. ago (DML and CG lithologies), similar to the crystallization ages obtained for Shergotty and Los Angeles. The FG lithology gave an age of ~177 m.y., with a subsequent cosmogenic correction modifying the age to 223 (±6) m.y. A study of Ar systematics by Korochantseva et al. (2009) resulted in a whole rock age of 200–250 m.y. for Zagami and ~400 m.y. for Shergotty. The isotopic and lithologic heterogeneity within Zagami is thought to be a result of magma mixing or brecciationThe formation of a breccia through a process by which rock fragments of of various types are recemented or fused together. Click on Term to Read More (Nyquist et al., 2010).
On the other hand, some believe that these chronometers do not reflect a recent crystallization on Mars, but instead, consider the young ages to represent a major impact resetting event associated with observed shock effects ranging from mosaicism to maskelynization of plagioclase feldspar, which are features consistent with a shock stageA petrographic assessment, using features observed in minerals grains, of the degree to which a meteorite has undergone shock metamorphism. The highest stage observed in 25% of the indicator grains is used to determine the stage. Also called "shock level". Click on Term to Read More of S4–5 (20–55 GPa). In support of such an event, studies of shergottites by Goresy et al. (2013) revealed evidence of pervasive melting (amounting to at least 23 vol%) of feldspar (producing maskelynite), pyrrhotite, titanomagnetite, and ilmenite, along with partial melting of clinopyroxene, as well as the presence of various high-pressure polymorphs; all of these features present a high probability that the radiometric ages have undergone shock-induced resetting.
It has been argued by some reseachers (e.g., Chen and Wasserburg [1986]; Bouvier et al. [2007, 2009]) that the Pb–Pb and Ar–Ar isochrons of shergottites represent an old crystallization age of ~4–4.5 b.y. However, the inferrence of an ancient crystallization age for Zagami and other shergottites, and the hypothesis that was advanced ascribing the young ages to isotopic resetting during a secondary impact event, has been shown to be inconsistent with the degree of shock-heating the meteorites have experienced. In other words, Zagami does not exhibit petrological features of post-shock heating to the degree which would cause a loss of an amount of 40Ar commensurate with an age of ~4 b.y. (Bogard and Park, 2007). Moreover, high degrees of shock would have led to chemical and isotopic equilibration of olivine and pyroxene, and the observed igneous zoning would have been obliterated (J. Jones, 2007); in fact, very little shock melt is present. Moreover, the concordant ages determined for the Re–Os, Rb–Sr, Lu–Hf, and Sm–Nd are inconsistent with shock resetting (Brandon, 2012). The isotopic anomalies observed in the Pb–Pb and Ar–Ar systems were considered instead to be the result of impact ejection (~30 GPa for Zagami with a temperature increase of ~70°C).
More recently it was asserted by J. H. Jones (2015) that the old ages calculated for shergottites based on the Pb–Pb system do not represent the shergottite crystallization age, since the Pb was likely not produced through in situ decay, but instead may have been inherited or incorporated after shergottite crystallization, or perhaps more plausibly, that it represents terrestrial contamination. They concluded that the old Ar-based ages are inaccurate due to inherited 40Ar, while others have attributed the old ages to inaccurate corrections for cosmogenic 36Ar. Therefore, the young shergottite crystallization ages based on Rb–Sr and Sm–Nd isotopic systems are generally considered to be the most accurate.
In their Ar–Ar analyses of Zagami minerals representing distinct magma sequences, Bogard and Park (2008) concluded that the excess 40Ar was neither the result of shock-implantation of martian atmospheric gas nor the remnant of in situ decay of 40K from a rock that crystallized 4.1 b.y. ago. The former scenario is inconsistent with the correlation that is observed by Korochantseva et al. (2009) between the atmospheric Ar component, which was trapped during a secondary event, and the radiogenic Ar component, which chronicles the time of crystallization. In the latter scenario, a higher 40Ar concentration in plagioclase than in pyroxene would be expected, which is not the case. Bogard and Park (2008) concluded that the excess 40Ar present throughout Zagami was likely inherited from the source magma or obtained by assimilation of K-rich crustal material during the early magma phase. Their finding that the 40Ar concentrations are the same among the various minerals in Zagami is thought to reflect the modulation of radiogenic gas through a pressure release mechanism as the magma rose from a depth >7 km toward the surface.
It was proposed by Bouvier et al. (2005) that the Rb–Sr ages of shergottites were reset through groundwater dissolution of phosphates within the martian rock. However, the Rb–Sr age was obtained after removal of the phosphates, and it was demonstrated by Nyquist et al. (2009) for the shergottite NWA 1460 that the Rb–Sr isochron as well as other isochrons result from trace element partitioning during igneous crystallization; they consider that these isochrons accurately date this petrogenetic stage.
Albarède et al. (2008) proposed that the resetting of radiogenic chronometers could have occurred as a result of extensive percolation of hot sulfate-rich fluids, and thus argue that the 4.1 b.y. crystallization age is the accurate age. However, no signs of such alteration are observed, and the distribution of radiogenic Ar present among different minerals is inconsistent with known diffusion mechanisms. Furthermore, the Sm–Nd data were shown to be unaffected by any such groundwater metasomatism processes, while a low-temperature alteration disturbance of the Pb–Pb chronometer was shown to be inconsistent with shergottite features.
Other investigations of in situ U–Pb were conducted on baddeleyiteA rare zirconium oxide (ZrO2) mineral, often formed as a shock-induced breakdown product of zircon. This mineral can be found in some lunar and martian meteorites. Click on Term to Read More (zirconium dioxide), a late-stage crystallization mineral that forms readily under conditions of high oxygen fugacityUsed to express the idealized partial pressure of a gas, in this case oxygen, in a nonideal mixture. Oxygen fugacity (ƒO2) is a measure of the partial pressure of gaseous oxygen that is available to react in a particular environment (e.g. protoplanetary disk, Earth's magma, an asteroid's regolith, etc.) and Click on Term to Read More (Herd et al., 2007, 2012; Misawa and Yamaguchi, 2007). It was found that U–Pb systematics remain undisturbed at shock pressures up to 57 GPa and temperatures up to 1300°C, and accurately reflect a very young age for Zagami and other shergottites of ~166 m.y. and 182.1 (±6.9) m.y. Further studies utilizing Rb–Sr, Sm–Nd, Ar–Ar, and U–Pb ages all show consistently young ages for Zagami of approximately 166 m.y. (Borg et al., 2005; Park and Bogard, 2007). An Ar–Ar age study by Korochantseva et al (2009) was complicated by excess Ar, but they determined an age for Zagami of 200–250 m.y. In support of a young crystallization age for the shergottites, Walton et al. (2008) propose that the bias for young ages determined for the majority of martian meteorites reflects the selective influence of crystalline, consolidated, unaltered, unbrecciated, and unweathered material in the ejection process. This type of rock is typically located in fresh volcanic terranes at higher elevations (lower atmospheric densityMass of an object divided by its volume. Density is a characteristic property of a substance (rock vs. ice, e.g.). Some substances (like gases) are easily compressible and have different densities depending on how much pressure is exerted upon them. The Sun is composed of compressible gases and is much Click on Term to Read More) which favors a successful impact ejection. A young age of formation for shergottites was also determined through isotopic studies conducted by Humayun et al. (2013) and Moser et al. (2013), a synopsis for which can be found in a PSRD article by G. Jeffrey Taylor: ‘The Importance of When‘, June 2014.
The age discrepancy of shergottites was addressed by Blinova and Herd (2009) in a study involving the petrogenesis of the depleted shergottites. They proposed a three-stage formation model in which the ancient age of 4.535 b.y. for the depleted shergottites, as shown by Pb–Pb and Rb–Sr data, corresponds to the initial crystallization of the magma ocean. The investigators envision that both a Deep Mantle Source and a more shallow Naklite Source were formed at that time. A partial meltingAn igneous process whereby rocks melt and the resulting magma is comprised of the remaining partially melted rock (sometimes called restite) and a liquid whose composition differs from the original rock. Partial melting occurs because nearly all rocks are made up of different minerals, each of which has a different melting Click on Term to Read More event affecting the NakhliteOne of the Martian SNC meteorites, nakhlites are basaltic cumulate clinopyroxenite rocks, and most all are comprised mainly of sub-calcic augite with approximately 10% Fe-rich olivine (giving the augite a green color) that are set in a very fine-grained matrix (mesostasis) comprised of plagioclase, K-feldspar, clinopyroxene, Fe-Ti oxide (Ti-magnetite), sulfide, Click on Term to Read More Source occurred 1.3 b.y. ago, leaving a slightly depleted Nakhlite Residue component; they designated this event the Nakhlite Event. About 500 m.y. after the Nakhlite Event, an ascending hot plume (and possibly many) brought the depleted Deep Mantle Source into contact with the Nakhlite Residue, producing the depleted shergottite parental source magma (for which Y-980459 is a close match to this parental melt composition). Differential mixing of the Deep Mantle Source and the Nakhlite Residue could have produced the various compositions of depleted shergottites, while the enriched and intermediate shergottites could have formed through lower degrees of partial melting of more volatile-rich material located at greater distances from the plume. Their model also explains the Sm–Nd isotopic signatures observed in shergottites, including the young ~1.3 b.y. age, and it provides an explanation for the various redoxOxidation and reduction together are called redox (reduction and oxidation) and generally characterized by the transfer of electrons between chemical species, like molecules, atoms or ions, where one species undergoes oxidation, a loss of electrons, while another species undergoes reduction, a gain of electrons. This transfer of electrons between reactants Click on Term to Read More conditions observed in the shergottite suite; i.e., enriched shergottites were formed by partial melting of volatile- and incompatible-element-bearing trapped liquid in the parental source magma.
The problem of anomalously old shergottite ages obtained through 40Ar/39Ar chronometry was investigated by Cassata and Borg (2016). They determined that previous cosmogenic correction techniques for 36Ar were not accurate, so a new technique was applied in which cosmogenic corrections are based on CRE age data for un-irradiated material, while step-wise production rate estimates are implemeted taking into account spatial variations in Ca and K. Using this new Ar–Ar dating method, they obtained young ages for two shergottites (NWA 4468 and NWA 2975) that are in accord with ages based on other radiometric chronometers. By comparison, application of the common cosmogenic correction techniques to the same two shergottites led to erroneously old CRE ages that correspond to anomalously old isochron ages.
A subsequent shock event, which occurred ~3 m.y. ago, probably represents the ejection of this rock from Mars. Studies of the impact-melt glass veins have identified fine-grained material that is enriched in a felsicOne of the two broad categories of silicate minerals, the other being mafic, based on the magnesium (Mg) and/or iron (Fe) content. Felsic indicates silicate minerals that are not predominantly comprised of Mg and/or Fe. The term is derived from feldspar + ic (having the character or form of). The Click on Term to Read More component and depleted in a mafic component (Rao and McKay, 2002), as well as having an elevated sulfur abundance (Rao et al, 1999) and possibly an elevated Pb abundance (Borg et al, 2005). It is proposed that this impact glass material represents martian soil that was mechanically fractionated through impact gardening and metasomatism processes, and was then incorporated into the melt phase.
The Mars ejection age for Zagami pyroxene based on 37Ar is 2.2–3.0 m.y., determined by adding together the cosmic ray exposure (CRE) age and the terrestrial age (Eugster et al., 2002). The ejection ages of several other basaltic shergottites, including Shergotty (3.0 ±0.3 m.y.), Los Angeles (3.0 ±0.3 m.y.), and QUE 94201 (2.8 ±0.3 m.y.), suggest that they all may have experienced a simultaneous ejection event on Mars. Other olivine-bearing shergottites, including Dar al Gani 476 (and pairings) and Sayh al Uhaymir 005 (and pairings), have very similar ejection ages of 1.24 (±0.12) m.y. and 1.0–1.4 m.y., respectively. Their similar CRE ages, bulk chemical compositions, 26Al concentrations, and textures, may represent a common magma source and ejection event from Mars. The shergottite EETA79001 has an ejection age of 0.73 (±0.15) m.y. and may be associated with the DaG 476 ejection event, or alternatively, it may represent a late breakup in space of the lherzolitic shergottite meteoroidSmall rocky or metallic object in orbit around the Sun (or another star). consistent with its content of lherzolitic-type xenocrysts. All of the lherzolitic meteorites found so far have ejection ages that closely coincide (3.8 ±0.7 to 4.7 ±0.5 m.y.), indicating a likely ejection from Mars ~1 m.y. before most of the basaltic shergottites, or possibly that they crystallized later at depth. Cosmic ray exposure ages have now been determined for many more martian meteorites and Mahajan (2015) compiled a chart based on the reported CRE ages for 53 of them (out of a total of ~80 known at that time). He concluded that together these 53 meteorites represent 10 distinct impact events which occurred 0.92 m.y., 2.12 m.y., 2.77 m.y., 4.05 m.y., 7.3 m.y., 9.6 m.y., 11.07 m.y., 12.27 m.y., 15 m.y., and 16.73 m.y.—see his chart here. In a subsequent review based on multiple criteria, Irving et al. (2017 [#2068]) made a new determination of the number of separate launch events associated with the known (101 at the time of their study) martian meteorites. They speculate that the number could be as few as twenty, and suggest that Zagami and at least 26 other enriched olivine-free mafic shergottites were probably ejected in a few separate impact events ~2–5 m.y. ago.
A study was made on Zagami in which calculations were made of shock veins in order to determine the duration of the shock wave at impact (~10 ms), and with that, the commensurate size of the impact craterCrater formed by high-speed impact of a meteoroid, asteroid, or comet on a solid surface. Craters are a common feature on most moons (an exception is Io), asteroids, and rocky planets, and range in size from a few cm to over 1,000 km across. There is a general morphological progression Click on Term to Read More that was formed. The results indicate a craterBowl-like depression ("crater" means "cup" in Latin) on the surface of a planet, moon, or asteroid. Craters range in size from a few centimeters to over 1,000 km across, and are mostly caused by impact or by volcanic activity, though some are due to cryovolcanism. Click on Term to Read More size of 1.5–5.0 km (Walton et al., 2008). Additionally, an excellent candidate for one of the source craters that led to the spallationThe formation of new nuclides by interactions of high-energy cosmic ray protons with target nuclei that commonly produce several smaller product nuclides. of the late-forming shergottites is the 10.1-km-diameter rayed crater Zunil, which is the most conspicuous crater located in the young, Amazonian-aged (1.8 billion years ago to the present day), lava-covered Cerberus Planum within Elysium Planitia. Other shergottites may have been ejected from the young terrain at the base of the Ceraunius Tholus volcano in the Tharsis province, or from similar young craters reflecting ejection events consistent with CRE ages of <18 m.y. Based on data from the Infrared Mineralogical Mapping Spectrometer aboard the Mars Express orbiter, the best fits mineralogically are in the older volcanic terrains of the southern highlands including Syrtis Major, Thaumasia Planum, and Herperia Planum (Ody et al., 2013). The specimen shown above is a 21.0 g cut fragment of the coarse-grained component from the NZ (normal Zagami) lithology containing oriented pigeonite crystals and a segment of a black glassy shock-melt vein at the top edge.
∗ Recent geochemical research on the martian basalts has led to new petrogenetic models and classification schemes.read more >>
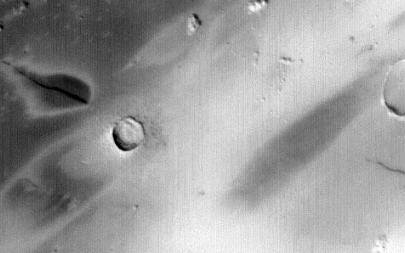
Photo courtesy of NASA/JPL/Malin Space Science Systems
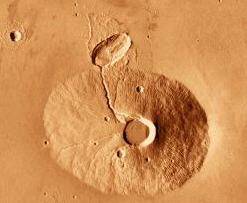

Photos Copyright © by Calvin J. Hamilton
A large oblique impact at the base of the Ceraunius Tholus volcano may have ejected material towards Earth.