Thermal Doppler Broadening
Spreading of a spectral line due to the temperature of the emitting medium. In a gas, the individual atoms, elements or molecules are continuously moving in random directions, with an average speed proportional to the temperature of the gas:
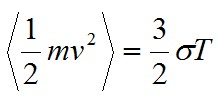
The term on the left-hand side is the mean kinetic energy. The individual gas particles (with mass m) follow a Maxwellian velocity distribution, resulting in a spread of velocities (v) about the average value. On the right hand side of the equation, T is the temperature of the gas and σ is the Stefan-Boltzmann constant.
At a given time, any particular atom, elementSubstance composed of atoms, each of which has the same atomic number (Z) and chemical properties. The chemical properties of an element are determined by the arrangement of the electrons in the various shells (specified by their quantum number) that surround the nucleus. In a neutral atom, the number of Click on Term to Read More or moleculeCollection of atoms held together by chemical bonds into a discrete, finite structure. One way molecules are represented is by a chemical formula where symbols for the elements are used to indicate the types of atoms present and subscripts are used to indicate the relative numbers of atoms. For example, Click on Term to Read More could be moving along the line-of-sight, perpendicular to the line-of-sight, or some combination of both. As a consequence, every spectral line emitted is Doppler shifted slightly higher or lower relative to the observer, broadening the spectral line. Thermal Doppler broadening is also possible for absorptionTransfer of energy to a medium as a particle or electromagnetic radiation passes through it. Absorption of electromagnetic radiation is the combined result of Compton scattering, σ, and photoelectric absorption, τ. It may be quantified: where, t = thickness, ρ = density, and μ = mass absorption coefficient, which combines Compton and photoelectric effects (μ = σ + τ). Click on Term to Read More lines, where particles in the absorbing medium have random motions, so Doppler shifts in the absorbed wavelengths can occur.

Imahe source: http://astronomy.swin.edu.au/cms/astro/cosmos/T/Thermal+Doppler+Broadening.






