NWA 1839
Meta-CO or -CV
(L7 in MetBull 89)

Found 2003
no coordinates recorded A 121.8 g meteoriteWork in progress. A solid natural object reaching a planet’s surface from interplanetary space. Solid portion of a meteoroid that survives its fall to Earth, or some other body. Meteorites are classified as stony meteorites, iron meteorites, and stony-iron meteorites. These groups are further divided according to their mineralogy and Click on Term to Read More was found in the Sahara Desert and later purchased by American collector N. Oakes in Rissani, Morocco in June 2003. This meteorite was analyzed at Northern Arizona University (T. Bunch and J. Wittke) and was initially determined to be a highly recrystallized L7 chondriteChondrites are the most common meteorites accounting for ~84% of falls. Chondrites are comprised mostly of Fe- and Mg-bearing silicate minerals (found in both chondrules and fine grained matrix), reduced Fe/Ni metal (found in various states like large blebs, small grains and/or even chondrule rims), and various refractory inclusions (such Click on Term to Read More. Notwithstanding this L7 classification, a subsequent oxygenElement that makes up 20.95 vol. % of the Earth's atmosphere at ground level, 89 wt. % of seawater and 46.6 wt. % (94 vol. %) of Earth's crust. It appears to be the third most abundant element in the universe (after H and He), but has an abundance only Click on Term to Read More isotopeOne of two or more atoms with the same atomic number (Z), but different mass (A). For example, hydrogen has three isotopes: 1H, 2H (deuterium), and 3H (tritium). Different isotopes of a given element have different numbers of neutrons in the nucleus. Click on Term to Read More study (D. Rumble III, CIW) showed that NWA 1839 plots on the Carbonaceous ChondriteCarbonaceous chondrites represent the most primitive rock samples of our solar system. This rare (less than 5% of all meteorite falls) class of meteorites are a time capsule from the earliest days in the formation of our solar system. They are divided into the following compositional groups that, other than Click on Term to Read More Anhydrous MineralInorganic substance that is (1) naturally occurring (but does not have a biologic or man-made origin) and formed by physical (not biological) forces with a (2) defined chemical composition of limited variation, has a (3) distinctive set of of physical properties including being a solid, and has a (4) homogeneous Click on Term to Read More (CCAM) line within the field of CO chondritesChondrites are the most common meteorites accounting for ~84% of falls. Chondrites are comprised mostly of Fe- and Mg-bearing silicate minerals (found in both chondrules and fine grained matrix), reduced Fe/Ni metal (found in various states like large blebs, small grains and/or even chondrule rims), and various refractory inclusions (such Click on Term to Read More.
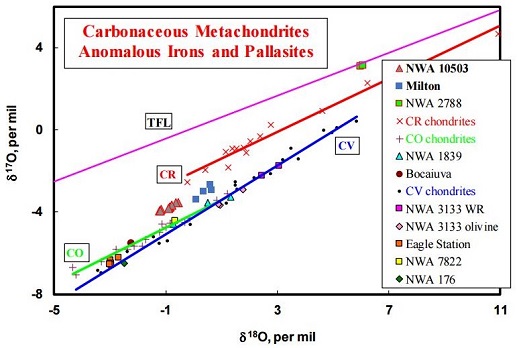
click on image for a magnified view Diagram credit: Irving et al., 79th MetSoc, #6461 (2016) Chromium vs. Oxygen Isotope Plot
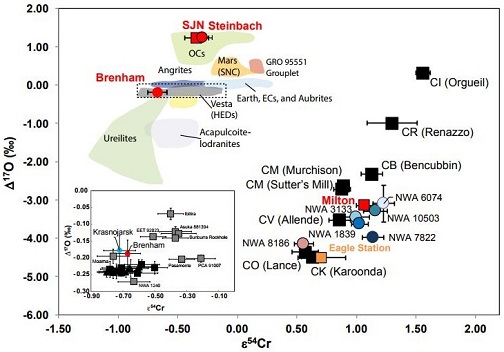
click on diagram for a magnified view Diagram credit: Sanborn et al., 49th LPSC, #1780 (2018) Northwest Africa 1839 is well preserved and exhibits only moderate weathering. It has an equigranular texture with no relict chondrulesRoughly spherical aggregate of coarse crystals formed from the rapid cooling and solidification of a melt at ~1400 ° C. Large numbers of chondrules are found in all chondrites except for the CI group of carbonaceous chondrites. Chondrules are typically 0.5-2 mm in diameter and are usually composed of olivine Click on Term to Read More visible. The low-Ca pyroxenes contain an abundance of CaO (Wo2.3), which by definition (see below) surpasses the maximum amount found in type 6 chondrites (ave. 1.3–1.8 mol%). Curiously, the high-Ca pyroxenes in NWA 1839 do not follow the defined CaO abundances of a type 7 chondrite—that is, a lower abundance for a higher petrologic typeMeasure of the degree of aqueous alteration (Types 1 and 2) and thermal metamorphism (Types 3-6) experienced by a chondritic meteorite. Type 3 chondrites are further subdivided into 3.0 through 3.9 subtypes.. Instead, in contrast to type 6 chondrites which have an average value of ~Wo45, NWA 1839 contains clinopyroxene with Wo47 (Mittlefehldt and Lindstrom, 2001). Northwest Africa 1839 exhibits a shock stageA petrographic assessment, using features observed in minerals grains, of the degree to which a meteorite has undergone shock metamorphism. The highest stage observed in 25% of the indicator grains is used to determine the stage. Also called "shock level". Click on Term to Read More of only S1, perhaps indicating a long period of annealing following peak shock effects. Type 7 ordinary chondrites were originally defined by Dodd et al. (1975) according to specific petrographic characteristics. They listed three metamorphic criteria to distinguish between petrographic types 6 and 7:
- the presence of poorly defined chondrules in type 6, but only relict chondrules in type 7
- low-Ca pyroxenes in type 6 contain no more than 1.0 wt% CaO (1.0 wt% = ~1.9 mol% Wo), but more than 1.0 wt% in type 7; conversely, the CaO content of high-Ca pyroxenes decreases from type 6 to type 7
- feldsparAn alumino-silicate mineral containing a solid solution of calcium, sodium and potassium. Over half the Earth’s crust is composed of feldspars and due to their abundance, feldspars are used in the classification of igneous rocks. A more complete explanation can be found on the feldspar group page. Click on Term to Read More grains gradually coarsen to reach a size of at least 0.1 mm in type 7
For those meteorites that experienced metamorphic temperatures high enough for metal–sulfide melting to occur, which most commonly occurs as a result of impact events, an igneous texture would be produced (Mittlefehldt and Lindstrom, 2001). In these cases the use of the Van Schmus–Wood classification scheme is no longer valid, and these meteorites could instead be referred to as primitive achondrites or metachondrites, or they might be impact melts.
Two views of a 1.43 g slice of NWA 1839 are shown above. The photo below shows a thin sectionThin slice or rock, usually 30 µm thick. Thin sections are used to study rocks with a petrographic microscope. viewed in partially polarized light in which the equigranular texture and absence of chondrules and metalElement that readily forms cations and has metallic bonds; sometimes said to be similar to a cation in a cloud of electrons. The metals are one of the three groups of elements as distinguished by their ionization and bonding properties, along with the metalloids and nonmetals. A diagonal line drawn Click on Term to Read More is evident.
Photo courtesy of T. Bunch—Northern Arizona University







